Tummy Tuck Trials Participation and Potential Compensation
Clinical trials for tummy tuck (abdominoplasty) procedures study new techniques, devices, or combinations of treatments to improve outcomes, reduce scarring, or speed recovery. Participating in a trial can provide access to cutting-edge care at reduced or no cost, while contributing to medical knowledge. Understanding how trials work, eligibility criteria, and potential risks and benefits helps you make an informed decision. Trials are conducted by academic medical centers, plastic surgery practices, and device manufacturers under FDA oversight.
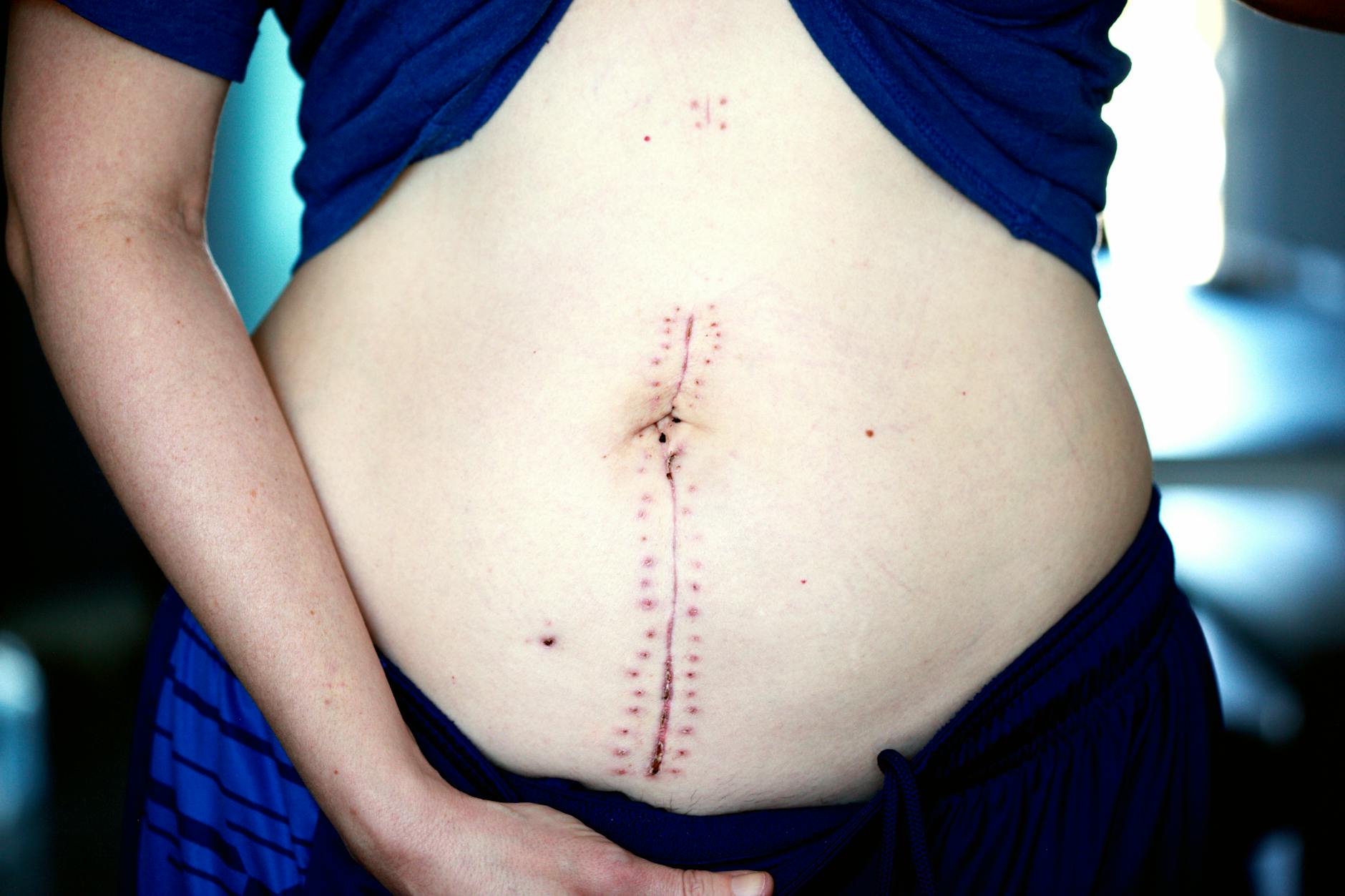
What Tummy Tuck Trials Study
Trials may evaluate new liposuction technologies, scar-reduction methods, pain management protocols, or combination procedures (e.g., tummy tuck with hernia repair). Some compare surgical techniques; others test non-surgical alternatives. Phase III trials—the final stage before FDA approval—typically involve established procedures with known safety profiles. Early-phase trials carry more uncertainty. Trials might study new suturing methods, drainage systems, or post-operative dressings. Some focus on patient-reported outcomes like satisfaction and quality of life. Reading the trial protocol will tell you exactly what is being tested and what you would receive.
Eligibility and Screening
Each trial has specific inclusion and exclusion criteria: age, BMI, health status, prior surgeries, and willingness to follow the protocol. You will undergo screening to determine eligibility. Be honest about your medical history—this protects you and the integrity of the research. Not everyone who applies will qualify. Common exclusions include smoking, uncontrolled diabetes, bleeding disorders, and recent weight loss. Some trials require you to be at a stable weight for 6–12 months. The screening process may include blood work, imaging, and a physical exam.
Risks and Benefits
Benefits may include free or discounted surgery, close monitoring by a research team, and access to innovative treatments. Risks include unknown side effects, the possibility of receiving a placebo (in some trial designs), and time commitments for follow-up visits. All trials must provide an informed consent document explaining the purpose, procedures, risks, and your rights. You can withdraw at any time. Compensation for participation varies—some trials offer no payment; others provide a stipend for time and travel. Ask about compensation before enrolling. Understand who pays for complications: the sponsor, your insurance, or you.
Finding and Evaluating Trials
ClinicalTrials.gov lists federally and privately funded trials. Search for "abdominoplasty" or "tummy tuck." Contact the trial coordinator for details. Ask about compensation for participation, who pays for complications, and what happens if you leave the trial. Consult your own doctor before enrolling to ensure the trial is appropriate for you. Check the principal investigator's credentials and the institution's reputation. Be wary of trials that seem too good to be true or that pressure you to enroll quickly.
Questions to Ask Before Enrolling
What phase is the trial, and what is known about the treatment's safety? What are the inclusion and exclusion criteria? How long will the trial last, and what is the time commitment for visits? Who pays for treatment, tests, and any complications? Can I continue my current medications? What happens if I withdraw? Will I receive the study drug or a placebo? Get answers in writing and discuss with your primary care provider. Do not feel pressured to enroll—participation is voluntary. Ask whether you will have access to the treatment after the trial ends if it helps you.
Weighing the Decision
Trials contribute to medical knowledge and may offer access to promising treatments. They also involve uncertainty and commitment. Consider your support system, ability to travel to the trial site, and comfort with the unknown. Some participants find meaning in contributing to research. Others prefer standard care with established outcomes. There is no wrong choice—only the one that fits your values and situation. If you have a specific timeline (e.g., a wedding), a trial's follow-up requirements may not fit. Discuss with your surgeon and family before deciding.
Post-Trial and Standard Care Options
If you choose not to participate in a trial or complete one, standard tummy tuck care remains an option. A board-certified plastic surgeon can perform abdominoplasty with proven techniques. Recovery typically involves 2–6 weeks of limited activity, compression garments, and follow-up visits. Results are permanent if you maintain a stable weight. Combine with liposuction for enhanced contouring. Discuss scarring expectations—incisions are typically placed low and can be hidden by underwear. Standard surgery costs $6,000–12,000 depending on complexity and location. Financing and payment plans are often available.
Taking the Next Step
Whether you pursue a clinical trial or standard surgery, the key is making an informed choice. Gather information from ClinicalTrials.gov, consult your primary care provider, and discuss options with a board-certified plastic surgeon. If you participate in a trial, follow the protocol exactly and report any concerns promptly. Keep copies of all consent documents. You can withdraw at any time without penalty. If you choose standard care, select a surgeon with experience in abdominoplasty and ask to see before-and-after photos. Your body, your choice—take the time to find the path that fits your goals and comfort level.